-
摘要: 高温胁迫下小麦光合能力下降是影响小麦灌浆和导致产量下降的主要因素。探明高温对小麦旗叶光合机构的伤害机制有助于小麦高温抗性资源的合理利用。本研究选取35个河南省不同年代的主栽小麦品种, 在离体条件下采用快速叶绿素荧光、820 nm光反射、延迟荧光同步测定的方法, 对不同温度(22 ℃, 30 ℃, 38 ℃)处理下所有小麦品种光合电子传递活性相关参数进行了测定和分析。首先根据最大光化学效率(FV/FM)对高温的敏感程度将35个小麦品种分为高温不敏感(G1)和高温敏感(G2)两种类型, 然后对所测参数分别平均, 结果显示高温胁迫下两种类型小麦品种光系统Ⅱ (PSⅡ)的光能捕获效率、吸收的光能推动电子传递到初级醌受体(QA)下游和光系统Ⅰ (PSⅠ)末端的效率、质体醌(PQ)库的再还原能力高温下均显著下降, 但G2小麦品种下降幅度更大; 两种类型小麦品种PSⅠ活性均不受影响, 但G2小麦品种PSⅠ供体侧电子传递活性下降幅度大于G1小麦品种。通过3种研究方法的相互补充和印证, 最终表明PSⅡ反应中心活性、PSⅡ光能的捕获和QA向下游传递电子的能力的差异是导致两种类型小麦品种光合电子传递活性差异的主要原因, PSⅡ供体侧和PSⅠ活性对小麦光系统Ⅱ高温抗性没有直接影响。该研究对未来冬小麦抗高温品种选育和栽培管理技术的创新提供了参考。Abstract: High temperatures are one of the main environmental stresses at the filling stage of winter wheat. The reduction in wheat photosynthesis caused by heat stress affects the filling of wheat and reduces grain yield. China is rich in wheat cultivars, and their photosynthetic sensitivity to high temperatures varies. Investigating the mechanism of high-temperature damage to the photosynthetic apparatus of wheat flag leaves can help to rationalize wheat high-temperature resistance resources. In this study, 35 wheat cultivars planted widely in Henan Province during different historical periods were selected. The parameters related to photosynthetic electron transfer of all wheat cultivars were measured and analyzed using fast chlorophyll fluorescence, 820 nm light reflection, and delayed fluorescence synchronization determination. First, according to the maximum photochemical efficiency (FV/FM) under high temperature, 35 wheat cultivars were divided into two groups: high-temperature insensitivity and high-temperature sensitivity, and the parameters measured for the two types of wheat cultivars were averaged. The results showed that J and I points of chlorophyll fluorescence induction curves were raised and the maximum quantum yield for primary photochemistry (
φPO), quantum yield for electron transport ( φEo), quantum yield for reduction of end electron acceptors at the PSⅠ acceptor side ( φRo), and performance index (PIABS) were significantly reduced in the two types of wheat cultivars under high-temperature stress. Moreover, the rise and decrease extents of high-temperature sensitive type were greater than those of high-temperature insensitive type, indicating that the PSⅡ light energy capture efficiency, the efficiency of the absorbed light energy to drive electrons downstream of the primary electron quinone acceptor QA, its efficiency to the PSⅠ end, and the re-reduction ability of the PQ pool decreased more in the leaves of the sensitive wheat cultivars under high temperatures. There was no significant decrease in the maximum decrease slope of the 820 nm light reflection curve (VPSⅠ) in either types of wheat cultivars, and the maximum increase slope of the 820 nm light reflection curve (VPSⅡ-PSⅠ) in both types of wheat cultivars significantly decreased. Furthermore, the decline of VPSⅡ-PSⅠ in the high-temperature sensitive wheat cultivars was greater than in the insensitive ones, indicating that the PSⅠ activity was not affected and the donor side of PSⅠ had a greater extent of damage in the high-temperature sensitive wheat cultivars. Values of both characteristic points of the delayed fluorescence induction curves (I1 and I2) decreased. At the same time, I2/I1 increased significantly at high temperatures and increased even more in high-temperature sensitive wheat cultivars, indicating that PSⅡ activity decreased and the efficiency of PSⅠ donor-side electron transfer to the end of PSⅠ increased. Based on the complementarity and confirmation of the three research methods, it is concluded that the difference in PSⅡ reaction center activity, PSⅡ light energy capture, and electron transfer from the acceptor side of QA to PSⅠ downstream is the main reason for the difference in photosynthetic ability between two types of wheat cultivars under high temperature. The PSⅡ donor side and PSⅠ activity have no direct impact on the high-temperature resistance of wheat. The results of this study help understand the current status of high-temperature resistance of wheat cultivars in the Huanghuai wheat-planting area and provide references for the breeding selection of high-temperature-resistant cultivars and innovation in cultivation techniques. -
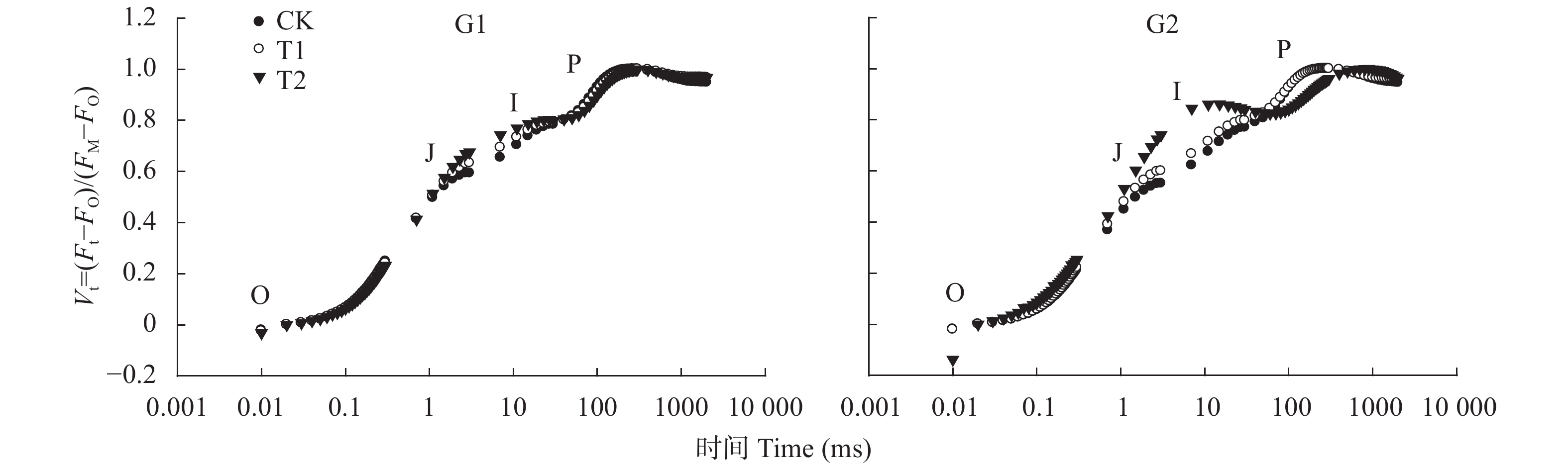
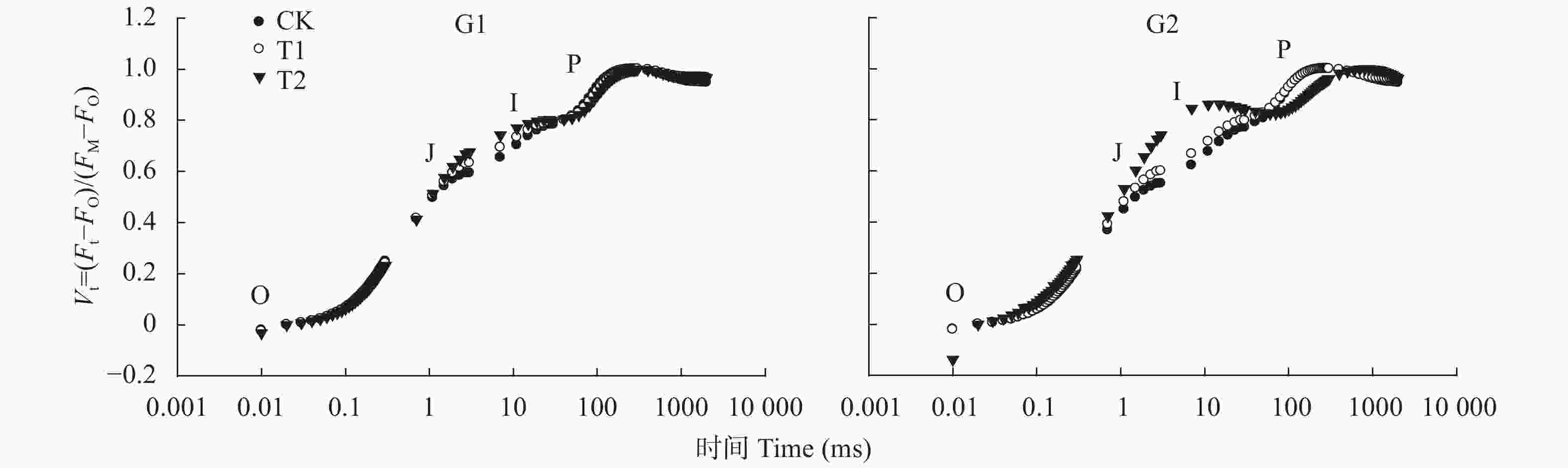
图 1 高温对高温不敏感型(G1)和高温敏感型(G2)小麦品种旗叶叶绿素荧光诱导曲线的影响(曲线已标准化)
CK、T1和T2表示叶片处理温度分别为22 ℃、30 ℃和38 ℃。O、J、I和P均为荧光诱导动力学曲线的特征位点。CK, T1 and T2 are treatments of 22 ℃, 30 ℃ and 38 ℃. O, J, I, and P are characteristic sites of the fluorescence induction curve.
Figure 1. Effect of high temperature on chlorophyll fluorescence induction curves of wheat flag leaves of high-temperature insensitive (G1) and sensitive (G2) cultivars (the curve has been standardized)
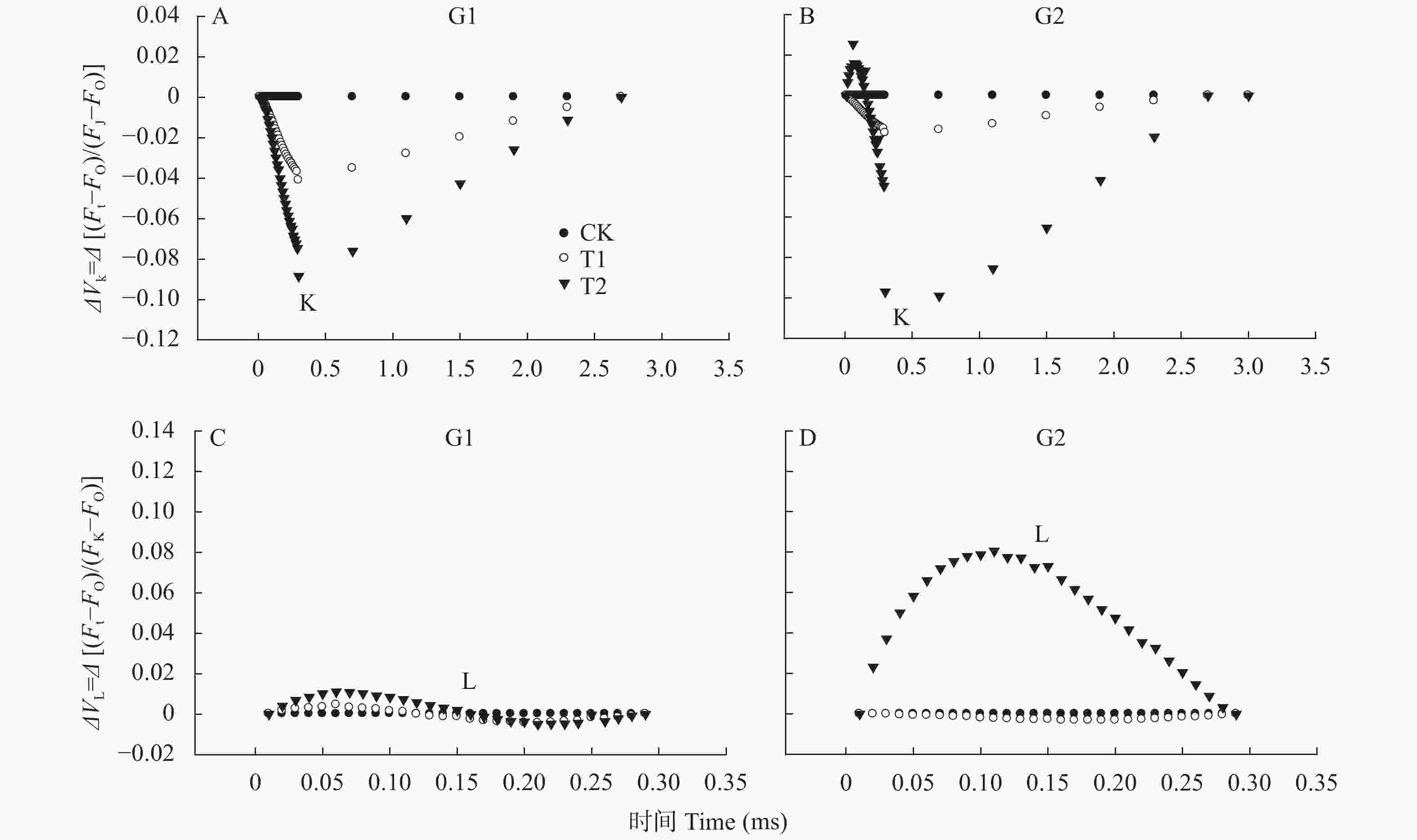
图 2 高温对高温不敏感型(G1)和高温敏感型(G2)小麦品种旗叶叶绿素荧光诱导曲线O-J段和O-K段荧光强度的影响
图A和B通过公式VK=(Ft−FO)/(FJ−FO)对O-J段荧光标准化, 然后使用公式ΔVK=VKtreatment−VKCK进行二次标准化; 图C和D通过公式VL=(Ft−FO)/(FK−FO)对O-K段荧光标准化, 然后使用公式ΔVL=VLtreatment−VLCK进行二次标准化。CK、T1和T2表示叶片处理温度分别为22 ℃、30 ℃和38 ℃。For fig. A and B, O-J segment were normalized by formula VK=(Ft −FO)/(FJ−FO), then ΔVK was calculate by formula ΔVK=VKtreatment−VKCK. For fig. C and D, O-K segment were normalized by formula VL=(Ft −FO)/(FK−FO), then ΔVL was calculate by formula ΔVL=VLtreatment−VLCK. CK, T1 and T2 are treatments of 22 ℃, 30 ℃ and 38 ℃, respectively.
Figure 2. Effect of high temperature on the fluorescence intensities of O-J segment and O-K segment of chlorophyll fluorescence induction curves of wheat flag leaves of high-temperature insensitive (G1) and sensitive (G2) cultivars
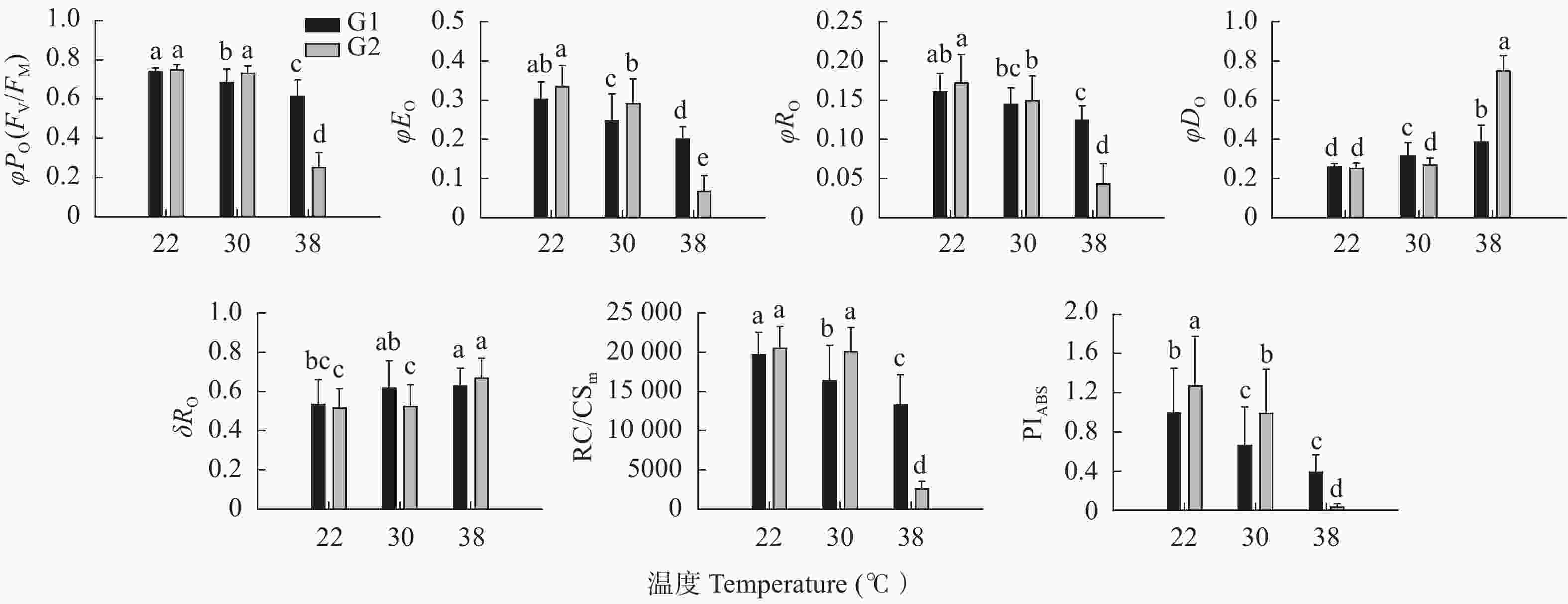
图 3 高温对高温不敏感型(G1)和高温敏感型(G2)小麦品种旗叶叶绿素荧光参数φPo、φEo、φRo、φDo、RC/CSm、δRo、PIABS 的影响
不同小写字母表示不同品种类型(G1和G2)不同温度间有显著差异(LSD, P<0.05), G1为13个小麦品种的平均值, G2为22个小麦品种的平均值。CK、T1和T2表示叶片处理温度分别为22 ℃、30 ℃和38 ℃。Different lowercase letters indicate significant differences among different temperatures of G1 and G2 (LSD, P<0.05). The value of G1 is the average of 13 wheat cultivars; the value G2 is the average of 22 wheat cultivars. CK, T1 and T2 are treatments of 22 ℃, 30 ℃ and 38 ℃, respectively.
Figure 3. Effect of high temperature on chlorophyll fluorescence parameters φPo, φEo, φRo, φDo, RC/CSm, δRo and PIABS of wheat flag leaves of high-temperature insensitive (G1) and sensitive (G2) cultivars
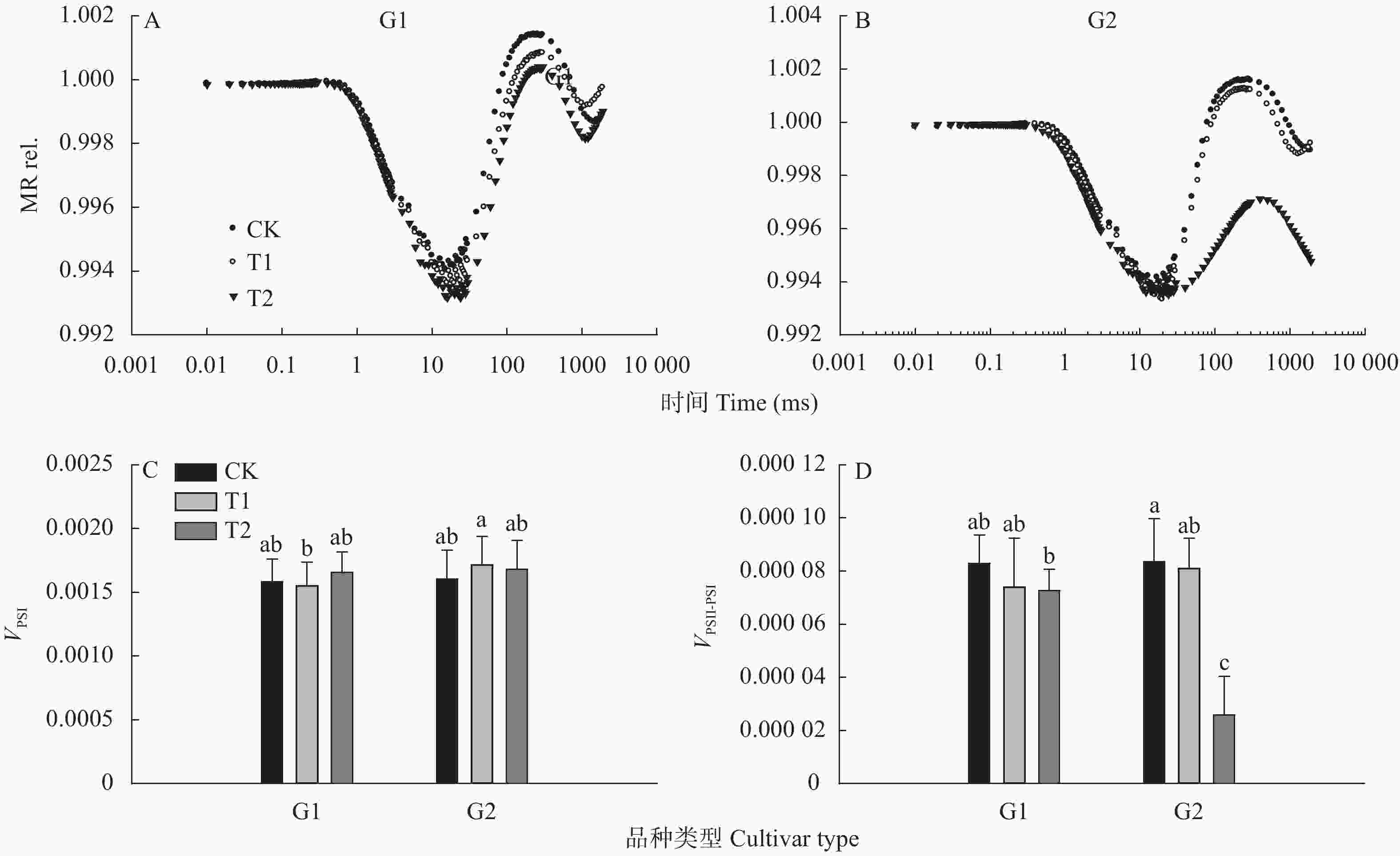
图 4 高温对高温不敏感型(G1)和高温敏感型(G2)小麦品种旗叶820 nm光反射(MR rel.)及其最大下降速率(VPSⅠ)和上升速率(VPSⅡ−PSⅠ)的影响
不同小写字母表示不同品种类型(G1和G2)不同处理间有显著差异(LSD, P<0.05), G1为13个小麦品种的平均值, G2为22个小麦品种的平均值。Different lowercase letters indicate significant differences among different temperatures of G1 and G2 (LSD, P<0.05). The value of G1 is the average of 13 wheat cultivars; G2 is the average of 22 wheat cultivars.
Figure 4. Effect of high temperature on the light reflection at MR820nm (MR rel.) and its maximum decrease rate (VPSⅠ) and rise rate (
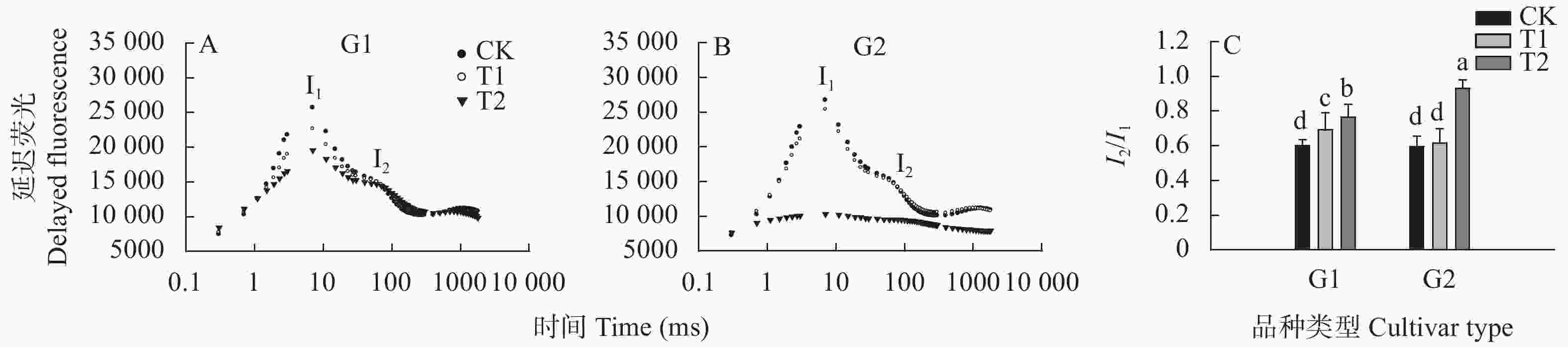
VPSⅡ−PSⅠ) of wheat flag leaves of high-temperature insensitive (G1) and sensitive (G2) cultivars 图 5 高温对高温不敏感型(G1)和高温敏感型(G2)小麦品种旗叶延迟荧光诱导曲线(0.01~0.02 ms) (A和B)和延迟荧光参数I2/I1 (C)的影响
不同小写字母表示不同品种类型(G1和G2)不同处理间有显著差异(LSD, P<0.05), G1为13个小麦品种的平均值, G2为22个小麦品种的平均值。I1和I2为延迟荧光诱导曲线的特征位点。Different letters indicate significant differences among different temperatures of G1 and G2 (LSD, P<0.05). The value of G1 is the average of 13 wheat cultivars; G2 is the average of 22 wheat cultivarss. I1 and I2 are characteristic sites of the delayed fluorescence induction curve.
Figure 5. Effect of high temperature on the delayed fluorescence induction curve (0.01−0.02 ms) (A and B) and delayed fluorescence parameter I2/I1 (C) of wheat flag leaves of high-temperature insensitive (G1) and sensitive (G2) cultivars
表 1 选用的小麦品种
Table 1. Wheat cultivars used in this study
编号
Number品种名称
Variety name编号
Number品种名称
Variety name1 碧玛1号 Bima 1 19 郑麦366 Zhengmai 366 2 碧玛4号 Bima 4 20 周麦22 Zhoumai 22 3 阿夫 Afu 21 郑麦7698 Zhengmai 7698 4 阿勃 Abo 22 矮丰3号 Aifeng 3 5 郑州7023 Zhengzhou 7023 23 郑麦0856 Zhengmai 0856 6 丰产3号 Fengchan 3 24 郑麦0943 Zhengmai 0943 7 郑州761 Zhengzhou 761 25 平原50 Pingyuan 50 8 西安8号 Xi’an 8 26 葫芦头 Hulutou 9 百农3217 Bainong 3217 27 大口麦 Dakoumai 10 宝丰7228 Baofeng 7228 28 红和尚头 Hongheshangtou 11 豫麦13 Yumai 13 29 西农6028 Xinong 6028 12 周麦9号 Zhoumai 9 30 白玉皮 Baiyupi 13 豫麦18 Yumai 18 31 偃展4110 Yanzhan 4110 14 豫麦49 Yumai 49 32 内乡5号 Neixiang 5 15 郑麦9023 Zhengmai 9023 33 博爱7023 Bo’ai 7023 16 兰考矮早8 Lankaoaizao 8 34 宛7107 Wan 7107 17 周麦18 Zhoumai 18 35 豫麦2号 Yumai2 18 矮抗58 Aikang 58 表 2 叶绿素快速荧光诱导动力学曲线参数及公式
Table 2. Parameters and formulas of rapid chlorophyll fluorescence inducing kinetic curve
参数
Parameter计算方式
Method of calculationFM 暗适应后照光获得的最大荧光强度 Maximum fluorescence intensity obtained under light after dark adaptation FO 叶绿素荧光诱导动力学曲线20 μs的荧光强度 Fluorescence intensity at 20 μs of rapid chlorophyll fluorescence inducing kinetic (JIP) curve FV FV=FM−FO Ft t时的荧光强度 Fluorescence intensity at t time FK K点(0.3 ms)的荧光强度 Fluorescence intensity at 0.3 ms FJ J点(3 ms)的荧光强度 Fluorescence intensity at 3 ms FI I点(30 ms)的荧光强度 Fluorescence intensity at 30 ms VJ VJ=(FJ−FO)/(FM−FO) VI VI=(FI−FO)/(FM−FO) MO MO=4(F300μs−FO)/(FM−FO) F300μs为300 μs的荧光强度 F 300μs is fluorescence intensity at 300 μs. ΨO ψO=1−VJ φPO φPO=Fv/FM=(FM−FO)/FM φEO φEo=ETo/ABS=[1−(FO/FM)]×ψO φDO φDO=1−φPO φRO φRO=φPO×(1−VI) σRO σRO=(1−VI)/(1−VJ) ABS/RC ABS/RC=MO×(1/VJ)×(1/φPO) RC/CSm RC/CSm=φPO×(VJ/MO)×FM PIABS PIABS=(RC/ABS)×[φPO/(1−φPO)]×[ψO/(1−ψO)] 表 3 不同类型小麦品种不同温度处理下的旗叶FV/FM及其38 ℃处理下的下降幅度和分类结果
Table 3. FV/FM of flag leaves at different temperatures and its decrease extents at 38 ℃, and classification results of different wheat cultivars
品种类型
Cultivar type品种编号
Cultivar number22 ℃ (CK) 30 ℃ (T1) 38 ℃ (T2) 38 ℃处理下降幅度
Decline extent at 38 ℃ (%)G1 22 0.723±0.035 0.724±0.019 0.713±0.019 1.40 G1 2 0.745±0.019 0.767±0.008 0.712±0.069 4.40 G1 23 0.764±0.018 0.674±0.023 0.720±0.022 5.80 G1 19 0.760±0.014 0.613±0.052 0.685±0.026 9.80 G1 17 0.749±0.010 0.710±0.205 0.649±0.056 13.40 G1 21 0.732±0.018 0.597±0.067 0.631±0.041 13.80 G1 20 0.739±0.045 0.539±0.071 0.615±0.065 16.80 G1 16 0.716±0.017 0.634±0.194 0.579±0.093 19.10 G1 18 0.759±0.016 0.739±0.023 0.603±0.141 20.60 G1 13 0.732±0.023 0.731±0.017 0.557±0.055 23.90 G1 15 0.765±0.021 0.731±0.021 0.574±0.066 25.00 G1 14 0.715±0.026 0.720±0.023 0.496±0.075 30.70 G1 4 0.732±0.031 0.731±0.022 0.443±0.06 39.50 G2 1 0.699±0.037 0.695±0.040 0.373±0.069 46.70 G2 7 0.757±0.012 0.724±0.027 0.381±0.058 49.70 G2 26 0.761±0.020 0.747±0.013 0.368±0.053 51.70 G2 3 0.753±0.021 0.737±0.030 0.351±0.086 53.40 G2 5 0.774±0.007 0.733±0.021 0.342±0.029 55.80 G2 27 0.771±0.014 0.754±0.017 0.324±0.036 58.00 G2 25 0.772±0.027 0.774±0.009 0.314±0.034 59.30 G2 6 0.763±0.036 0.715±0.020 0.277±0.077 63.60 G2 9 0.679±0.045 0.698±0.016 0.222±0.021 67.20 G2 33 0.712±0.037 0.756±0.012 0.225±0.033 68.40 G2 10 0.736±0.032 0.611±0.043 0.231±0.019 68.60 G2 34 0.775±0.009 0.759±0.009 0.244±0.070 68.60 G2 31 0.713±0.050 0.758±0.01 0.218±0.017 69.40 G2 35 0.785±0.031 0.747±0.031 0.240±0.044 69.40 G2 28 0.730±0.017 0.774±0.01 0.199±0.029 72.80 G2 24 0.767±0.010 0.752±0.009 0.195±0.013 74.50 G2 11 0.731±0.010 0.691±0.022 0.183±0.014 75.00 G2 8 0.735±0.024 0.725±0.014 0.183±0.031 75.10 G2 30 0.743±0.043 0.760±0.013 0.180±0.031 75.70 G2 32 0.762±0.040 0.758±0.014 0.172±0.028 77.40 G2 29 0.768±0.004 0.726±0.013 0.161±0.023 79.00 G2 12 0.761±0.009 0.700±0.024 0.130±0.038 82.90 G1为高温不敏感小麦类型, G2为高温敏感小麦类型。G1 represents the high temperature insensitive wheat cultivars and G2 represents the high temperature sensitive wheat cultivars. -
[1] 蔡剑, 姜东. 气候变化对中国冬小麦生产的影响[J]. 农业环境科学学报, 2011, 30(9): 1726−1733CAI J, JIANG D. The effect of climate change on winter wheat production in China[J]. Journal of Agro-Environment Science, 2011, 30(9): 1726−1733 [2] ASSENG S, FOSTER I, TURNER N C. The impact of temperature variability on wheat yields[J]. Global Change Biology, 2011, 17(2): 997−1012 doi: 10.1111/j.1365-2486.2010.02262.x [3] DJANAGUIRAMAN M, NARAYANAN S, ERDAYANI E, et al. Effects of high temperature stress during anthesis and grain filling periods on photosynthesis, lipids and grain yield in wheat[J]. BMC Plant Biology, 2020, 20(1): 268 doi: 10.1186/s12870-020-02479-0 [4] ZHAO C, LIU B, PIAO S L, et al. Temperature increase reduces global yields of major crops in four independent estimates[J]. Proceedings of the National Academy of Sciences of the United States of America, 2017, 114(35): 9326−9331 doi: 10.1073/pnas.1701762114 [5] CORREIA P M P, DA SILVA A B, ROITSCH T, et al. Photoprotection and optimization of sucrose usage contribute to faster recovery of photosynthesis after water deficit at high temperatures in wheat[J]. Physiologia Plantarum, 2021, 172(2): 615−628 doi: 10.1111/ppl.13227 [6] DEMIREVSKA-KEPOVA K, FELLER U. Heat sensitivity of Rubisco, Rubisco activase and Rubisco binding protein in higher plants[J]. Acta Physiologiae Plantarum, 2004, 26(1): 103−114 doi: 10.1007/s11738-004-0050-7 [7] CRAFTS-BRANDNER S, LAW R. Effect of heat stress on the inhibition and recovery of the ribulose-1, 5-bisphosphate carboxylase/oxygenase activation state[J]. Planta, 2000, 212(1): 67−74 doi: 10.1007/s004250000364 [8] DEGEN G E, ORR D J, CARMO-SILVA E. Heat-induced changes in the abundance of wheat rubisco activase isoforms[J]. New Phytologist, 2021, 229(3): 1298−1311 doi: 10.1111/nph.16937 [9] ENAMI I, KITAMURA M, TOMO T, et al. Is the primary cause of thermal inactivation of oxygen evolution in spinach PSⅡ membranes release of the extrinsic 33 kDa protein or of Mn?[J]. Biochimica et Biophysica Acta (BBA)—Bioenergetics, 1994, 1186(1/2): 52−58 [10] NARAYANAN S, TAMURA P J, ROTH M R, et al. Wheat leaf lipids during heat stress: Ⅰ. High day and night temperatures result in major lipid alterations[J]. Plant, Cell & Environment, 2016, 39(4): 787−803 [11] DJANAGUIRAMAN M, BOYLE D L, WELTI R, et al. Decreased photosynthetic rate under high temperature in wheat is due to lipid desaturation, oxidation, acylation, and damage of organelles[J]. BMC Plant Biology, 2018, 18(1): 55 doi: 10.1186/s12870-018-1263-z [12] FAN X L, ZHANG Z S, GAO H Y, et al. Photoinhibition-like damage to the photosynthetic apparatus in plant leaves induced by submergence treatment in the dark[J]. PLoS One, 2014, 9(2): e89067 doi: 10.1371/journal.pone.0089067 [13] YANG C, ZHANG Z S, GAO H Y, et al. The mechanism by which NaCl treatment alleviates psi photoinhibition under chilling-light treatment[J]. Journal of Photochemistry and Photobiology B: Biology, 2014, 140: 286−291 doi: 10.1016/j.jphotobiol.2014.08.012 [14] TÓTH S Z, SCHANSKER G, STRASSER R J. In intact leaves, the maximum fluorescence level (Fm) is independent of the redox state of the plastoquinone pool: a DCMU-inhibition study[J]. Biochimica et Biophysica Acta (BBA)—Bioenergetics, 2005, 1708(2): 275−282 doi: 10.1016/j.bbabio.2005.03.012 [15] SCHANSKER G, TÓTH S Z, STRASSER R J. Methylviologen and dibromothymoquinone treatments of pea leaves reveal the role of photosystem Ⅰ in the chl a fluorescence rise OJIP[J]. Biochimica et Biophysica Acta (BBA)—Bioenergetics, 2005, 1706(3): 250−261 [16] GAO J, LI P M, MA F W, et al. Photosynthetic performance during leaf expansion in Malus micromalus probed by chlorophyll a fluorescence and modulated 820 nm reflection[J]. Journal of Photochemistry and Photobiology B: Biology, 2014, 137: 144−150 doi: 10.1016/j.jphotobiol.2013.12.005 [17] ZUSHI K, MATSUZOE N. Using of chlorophyll a fluorescence OJIP transients for sensing salt stress in the leaves and fruits of tomato[J]. Scientia Horticulturae, 2017, 219: 216−221 doi: 10.1016/j.scienta.2017.03.016 [18] GUO Y J, LU Y P, GOLTSEV V, et al. Comparative effect of tenuazonic acid, diuron, bentazone, dibromothymoquinone and methyl viologen on the kinetics of chl a fluorescence rise OJIP and the MR820 signal[J]. Plant Physiology and Biochemistry, 2020, 156: 39−48 doi: 10.1016/j.plaphy.2020.08.044 [19] 杨程, 李鹏民, 张子山, 等. 叶绿素延迟荧光的发生及其在光合作用研究中的应用[J]. 植物生理学报, 2013, 49(12): 1277−1285YANG C, LI P M, ZHANG Z S, et al. Arising of chlorophyll delayed fluorescence and its application in photosynthesis research[J]. Plant Physiology Journal, 2013, 49(12): 1277−1285 [20] STRASSER R J, TSIMILLI-MICHAEL M, QIANG S, et al. Simultaneous in vivo recording of prompt and delayed fluorescence and 820-nm reflection changes during drying and after rehydration of the resurrection plant Haberlea rhodopensis[J]. Biochimica et Biophysica Acta (BBA)—Bioenergetics, 2010, 1797(6/7): 1313−1326 [21] SALVATORI E, FUSARO L, GOTTARDINI E, et al. Plant stress analysis: application of prompt, delayed chlorophyll fluorescence and 820 nm modulated reflectance. Insights from independent experiments[J]. Plant Physiology and Biochemistry, 2014, 85: 105−113 doi: 10.1016/j.plaphy.2014.11.002 [22] OUKARROUM A, GOLTSEV V, STRASSER R J. Temperature effects on pea plants probed by simultaneous measurements of the kinetics of prompt fluorescence, delayed fluorescence and modulated 820 nm reflection[J]. PLoS One, 2013, 8(3): e59433 doi: 10.1371/journal.pone.0059433 [23] CHEN W Y, JIA B, CHEN J Y, et al. Effects of different planting densities on photosynthesis in maize determined via prompt fluorescence, delayed fluorescence and P700 signals[J]. Plants (Basel, Switzerland), 2021, 10(2): 276 [24] ZHOU R H, KAN X, CHEN J J, et al. Drought-induced changes in photosynthetic electron transport in maize probed by prompt fluorescence, delayed fluorescence, P700 and cyclic electron flow signals[J]. Environmental and Experimental Botany, 2019, 158: 51−62 doi: 10.1016/j.envexpbot.2018.11.005 [25] 杨程, 张德奇, 杜思梦, 等. 黑暗诱导衰老对不同年代冬小麦品种旗叶光系统Ⅱ功能的影响[J]. 应用生态学报, 2018, 29(8): 2525−2531YANG C, ZHANG D Q, DU S M, et al. Effects of dark induced senescence on the function of photosystem Ⅱ in flag leaves of winter wheat released in different years[J]. Chinese Journal of Applied Ecology, 2018, 29(8): 2525−2531 [26] LIU M J, ZHANG Z S, GAO H Y, et al. Effect of leaf dehydration duration and dehydration degree on PSⅡ photochemical activity of Papaya leaves[J]. Plant Physiology and Biochemistry, 2014, 82: 85−88 doi: 10.1016/j.plaphy.2014.05.003 [27] 李鹏民, 高辉远, Reto J. Strasser. 快速叶绿素荧光诱导动力学分析在光合作用研究中的应用[J]. 植物生理与分子生物学学报, 2005, 31(6): 559−566LI P M, GAO H Y, STRASSER R J. Application of the fast chlorophyll fluorescence induction dynamics analysis in photosynthesis study[J]. Acta Photophysiologica Sinica, 2005, 31(6): 559−566 [28] 张菂, 陈昌盛, 李鹏民, 等. 利用快速荧光、延迟荧光和820 nm光反射同步测量技术探讨干旱对平邑甜茶叶片光合机构的伤害机制[J]. 植物生理学报, 2013, 49(6): 551−560ZHANG D, CHEN C S, LI P M, et al. Effects of drought on the photosynthetic apparatus in Malus hupehensis leaves explored by simultaneous measurement of prompt fluorescence, delayed fluorescence and modulated light reflection at 820 nm[J]. Plant Physiology Journal, 2013, 49(6): 551−560 [29] 金立桥, 车兴凯, 张子山, 等. 高温、强光下黄瓜叶片PSⅡ供体侧和受体侧的伤害程度与快速荧光参数Wk变化的关系[J]. 植物生理学报, 2015, 51(6): 969−976JIN L Q, CHE X K, ZHANG Z S, et al. The relationship between the changes in Wk and different damage degree of PSⅡ donor side and acceptor side under high temperature with high light in cucumber[J]. Plant Physiology Journal, 2015, 51(6): 969−976 [30] 张会玲, Marian Brestic, Katarina Olsovska, 等. 高温胁迫下不同热敏感性小麦光化学活性和能量分配差异[J]. 植物生理学报, 2015, 51(7): 1142−1150ZHANG H L, BRESTIC M, OLSOVSKA K, et al. Photochemical activity and energy distribution on wheat varieties with different heat-sensitivity under high temperature stress[J]. Plant Physiology Journal, 2015, 51(7): 1142−1150 [31] LI P M, CHENG L L, GAO H Y, et al. Heterogeneous behavior of PSⅡ in soybean (Glycine max) leaves with identical PSⅡ photochemistry efficiency under different high temperature treatments[J]. Journal of Plant Physiology, 2009, 166(15): 1607−1615 doi: 10.1016/j.jplph.2009.04.013 [32] NISHIYAMA Y, ALLAKHVERDIEV S I, MURATA N. A new paradigm for the action of reactive oxygen species in the photoinhibition of photosystem Ⅱ[J]. Biochimica et Biophysica Acta (BBA)—Bioenergetics, 2006, 1757(7): 742−749 doi: 10.1016/j.bbabio.2006.05.013 [33] CHEN S G, ZHOU F Y, YIN C Y, et al. Application of fast chlorophyll a fluorescence kinetics to probe action target of 3-acetyl-5-isopropyltetramic acid[J]. Environmental and Experimental Botany, 2011, 73: 31−41 doi: 10.1016/j.envexpbot.2011.08.005 [34] KAN X, REN J J, CHEN T T, et al. Effects of salinity on photosynthesis in maize probed by prompt fluorescence, delayed fluorescence and P700 signals[J]. Environmental and Experimental Botany, 2017, 140: 56−64 doi: 10.1016/j.envexpbot.2017.05.019 [35] SUN Y J, GAO Y L, WANG H, et al. Stimulation of cyclic electron flow around PSⅠ as a response to the combined stress of high light and high temperature in grape leaves[J]. Functional Plant Biology: FPB, 2018, 45(10): 1038−1045 doi: 10.1071/FP17269 -





 下载:
下载: