Distribution characteristics of soil fungi community in rhizosphere of foxtail millet under different planting patterns
-
摘要: 为了解谷子连作对土壤真菌群落结构的影响, 以撂荒地为对照, 以谷子-玉米轮作、谷子连作3年、连作5年根际土壤为研究对象, 采用真菌ITS高通量测序技术, 探究不同种植模式下谷子土壤真菌群落分布特征。结果表明: 不同种植模式下, 谷子根际土壤共检测到真菌10门24纲46目79科136属和146种。在门和纲水平上群体结构相对稳定, 谷子田土壤优势门主要包括子囊菌门和担子菌门, 优势纲为粪壳菌纲、座囊菌纲和盘菌纲, 在目水平谷子根际土壤粪壳菌目相对丰度是撂荒地的2倍以上, 在科水平和属水平轮作土壤被孢霉、球腔菌相对丰度高于连作土壤, 链格孢菌、亚隔孢壳菌和粉红螺旋聚孢霉菌相对丰度低于连作土壤。Alpha多样性分析显示, 谷子-玉米轮作与谷子连作根际土壤真菌丰度差异达显著水平(P<0.05), 轮作土壤真菌丰度最高。Beta多样性分析显示连作3年和连作5年根际土壤真菌结构相似, 与撂荒地以及轮作根际土壤真菌结构存在差异, 表明不同种植模式谷子根际土壤真菌群落结构发生了改变。相关性分析显示, 碱解氮与有机质呈极显著正相关(P<0.01), 与有效磷、脲酶活性呈显著相关(P<0.05), 多酚氧化酶活性与速效钾呈显著正相关(P<0.05)Chao1指数、Observed species指数与多酚氧化酶活性呈极显著正相关(P<0.01)。冗余分析(RDA)表明, CK受毛壳菌影响, CR受球腔菌属和微结节霉属的影响, TC和FC受毛葡孢属、毛喙壳属、亚隔孢壳属等影响。LEfSe分析确定了谷子根际土壤特定标志物, 轮作根际土壤的标志物包含被孢霉属和球腔菌属, 连作3年根际土壤标志物包含毛葡孢属、亚隔孢壳属和粉红螺旋聚孢霉属, 连作5年根际土壤标志物包含链格孢菌属和亚隔孢壳属。因此, 谷子-玉米轮作与谷子连作相比, 土壤真菌群落结构差异较大, 轮作土壤腐生菌较多, 连作土壤病原菌较多, 为谷子连作障碍研究提供了有用信息。Abstract: There are continuous cropping obstales in foxtail millet. To understand the effects of continuous cropping of foxtail millet on soil fungal community structure, we explored the distribution characteristics of fungal community of the rhizosphere soil using the fungal ITS high-throughput sequencing technology under different cropping strategy, including foxtail millet - maize rotation, foxtail millet continuous cropping for 3 years, and foxtail millet continuous cropping for 5 years, and the abandoned land taken as control. The results showed that a total of 10 phyla, 24 classes, 46 orders, 79 families, 136 genera and 146 species of fungi were detected in the rhizosphere soil of foxtail millet under different cropping strategy. At the phylum and class level, the population structure was relatively stable. The dominant phylum in soil mainly consisted of Ascomycota and Basidiomycota, while the dominant classes were Sordariomycetes, Dothideomycetes and Pezizomycetes. At the order level, the relative abundance of Sordariales in the rhizosphere of foxtail millet was 2 times more than that in the abandoned land. At family and genus level, the relative abundance of Mortierellaceae and Mycosphaerellaceae was higher, while the relative abundance of Alternaria, Didymella and Clonostachys was lower in rotation soil compared to continuous cropping soil. Alpha diversity analysis exhibited that the fungal abundance of the rhizosphere soil was of significant difference under foxtail millet - maize rotation and foxtail millet continuous cropping (P<0.05), and that under the rotation soil was the highest. Beta diversity analysis revealed that the fungal structures of the rhizosphere soil under continuous cropping for 3 years and 5 years were similar, and they were of difference from that under the abandoned cropping or the rotating cropping, indicating that the fungal community structure in the rhizosphere soil of foxtail millet was changed under different cropping strategy. Correlation analysis showed that Alkali-hydrolyzed nitrogen was significantly positive correlated with organic matter (P<0.01), and significantly correlated with available phosphorus and urease activities (P<0.05), while the activity of polyphenol oxidase was positive correlated with available potassium (P<0.05), and significantly positive correlated with Chao1 index and Observed species index(P<0.01). Redundancy analysis (RDA) indicated that CK, CR, TC and FC were affected by Chaetomium, Mycosphaerella and Microdochium, Botryotrichum, Chaetomidium and Didymella, respectively. LEfSe analysis identified distinctly specific markers of the rhizosphere soil of foxtail millet under different cropping strategy. The markers of rhizosphere soil contained Mortierella and Mycosphaerella for the rotating cropping, Botryotrichum, Didymella and Clonostachys for 3 years of continuous cropping Alternaria and Didymella,for 5 years of continuous cropping, respectively. All in all, the soil fungal community structure under millet - maize rotation croping, exhibiting more saprophytic bacteria and less pathogenic bacteria, was significantly different from that under foxtail millet continuous cropping, which provided useful information for the study on the continuous cropping obstacles of foxtail millet.
-
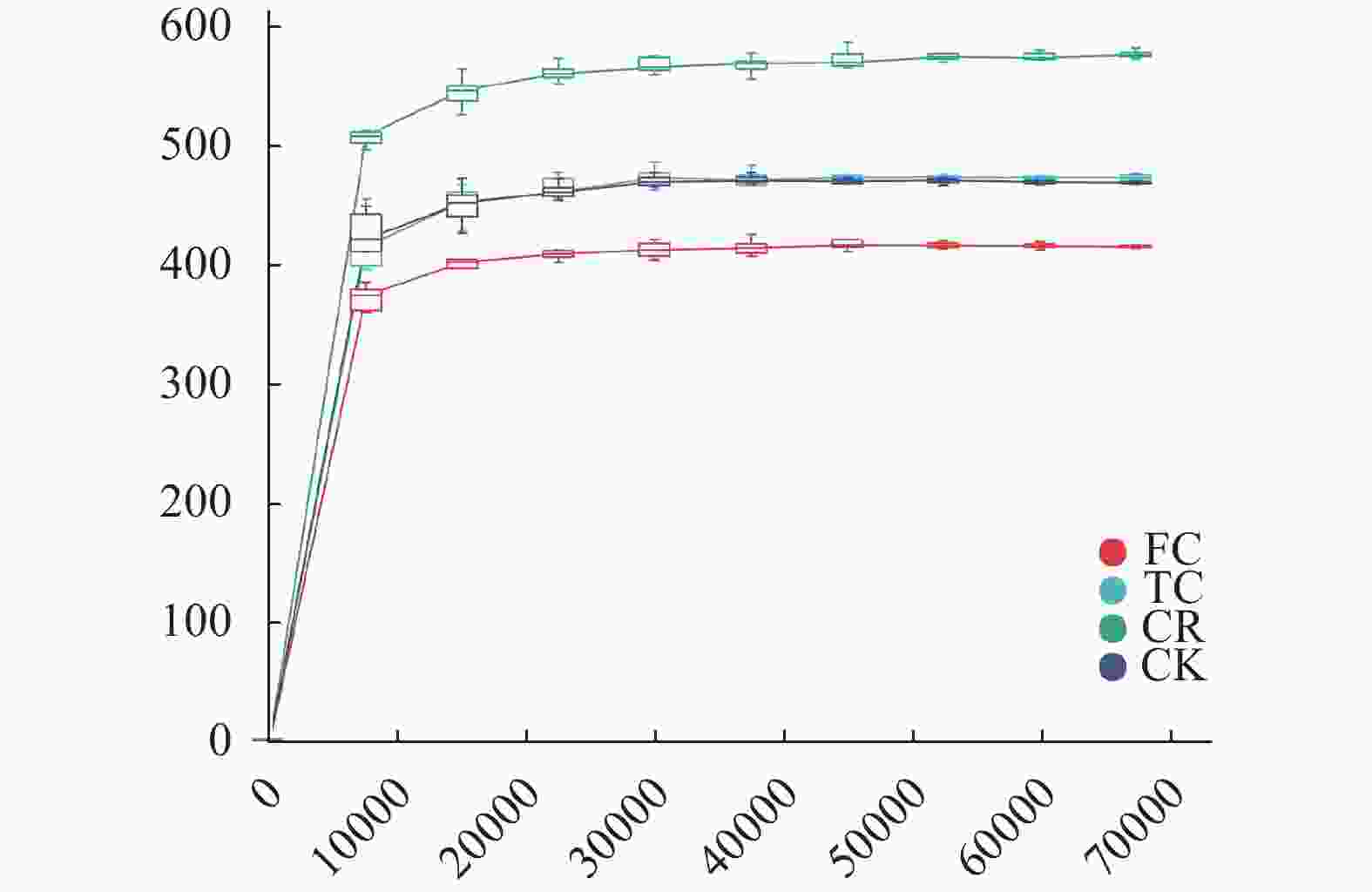
图 1 不同种植模式谷子土壤真菌群落的稀疏曲线
CK: 撂荒地; CR: 谷子-玉米轮作; TC: 谷子连作3年; FC: 谷子连作5年。CK: abandoned land; CR: foxtail millet rotation with maize; TC: continuous cropping for 3 years of foxtail millet; FC: continuous cropping for 5 years of foxtail mille.
Figure 1. Sparse curves of soil fungal community of Foxtail millet with different cropping patterns
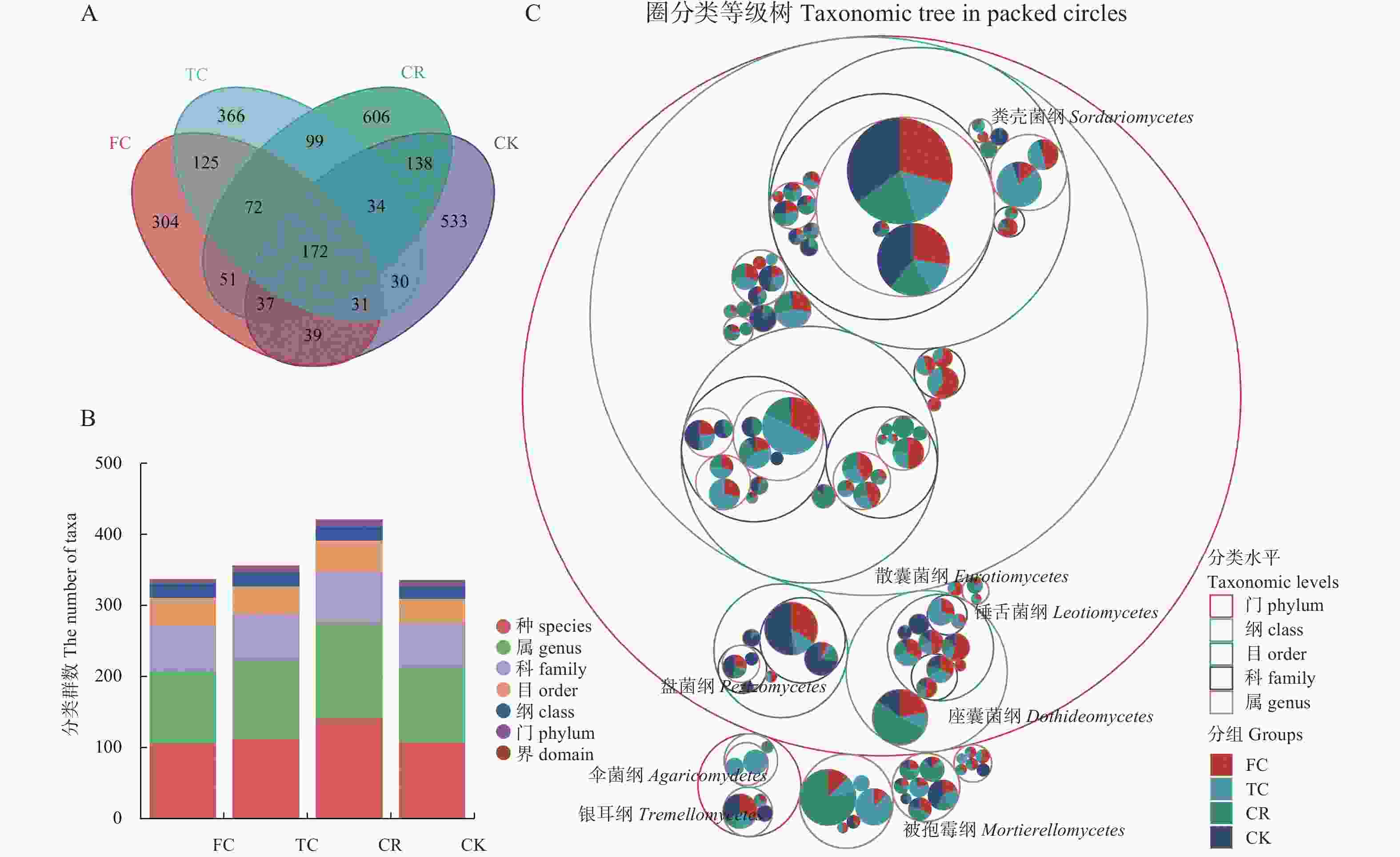
图 2 不同种植模式谷子土壤真菌ITS序列(ASV)分类
A: 不同种植模式ASV分布Venn图; B: 不同分类级别下不同种植模式ASV数; C: 不同种植模式谷子土壤真菌圈堆积图。CK: 撂荒地; CR: 谷子-玉米轮作; TC: 谷子连作3年; FC: 谷子连作5年。图C中, 在绘制土壤细菌分类等级树图的前提下, 将每个ASV的分组的丰度数据以饼图的形式添加到了图中。展示了谷子根际土壤真菌群落分类学构成, 最大的圈代表门水平, 逐渐缩小的圈按照梯度依次代表纲、目、科、属和种, 最内层圆点面积代表ASV的丰度大小, 同时也表示该ASV在各组中的组成比例。A: Venn diagram of ASV of different cropping patterns; B: ASVs number of different plant patterns at different classification levels; C: Taxonomic Tree in Packed Circles of soil fungi classification of foxtail millet under different cropping patterns. CK: abandoned land; CR: foxtail millet rotation with maize; TC: continuous cropping for 3 years of foxtail millet; FC: continuous cropping for 5 years of foxtail mille. For the figure C, on the premise of drawing the tree map of soil fungi classification, the abundance of each ASV group was added to the map in the form of pie chart. The largest circle represents phylum level, and the gradually shrinking circle represents class, order, family, genus and species according to the gradient. The innermost dot area represents the abundance of ASV, and also represents the composition proportion of ASV in each group.
Figure 2. Amplicon sequence variants (ASV) classification of soil fungi of foxtail millet in different planting patterns
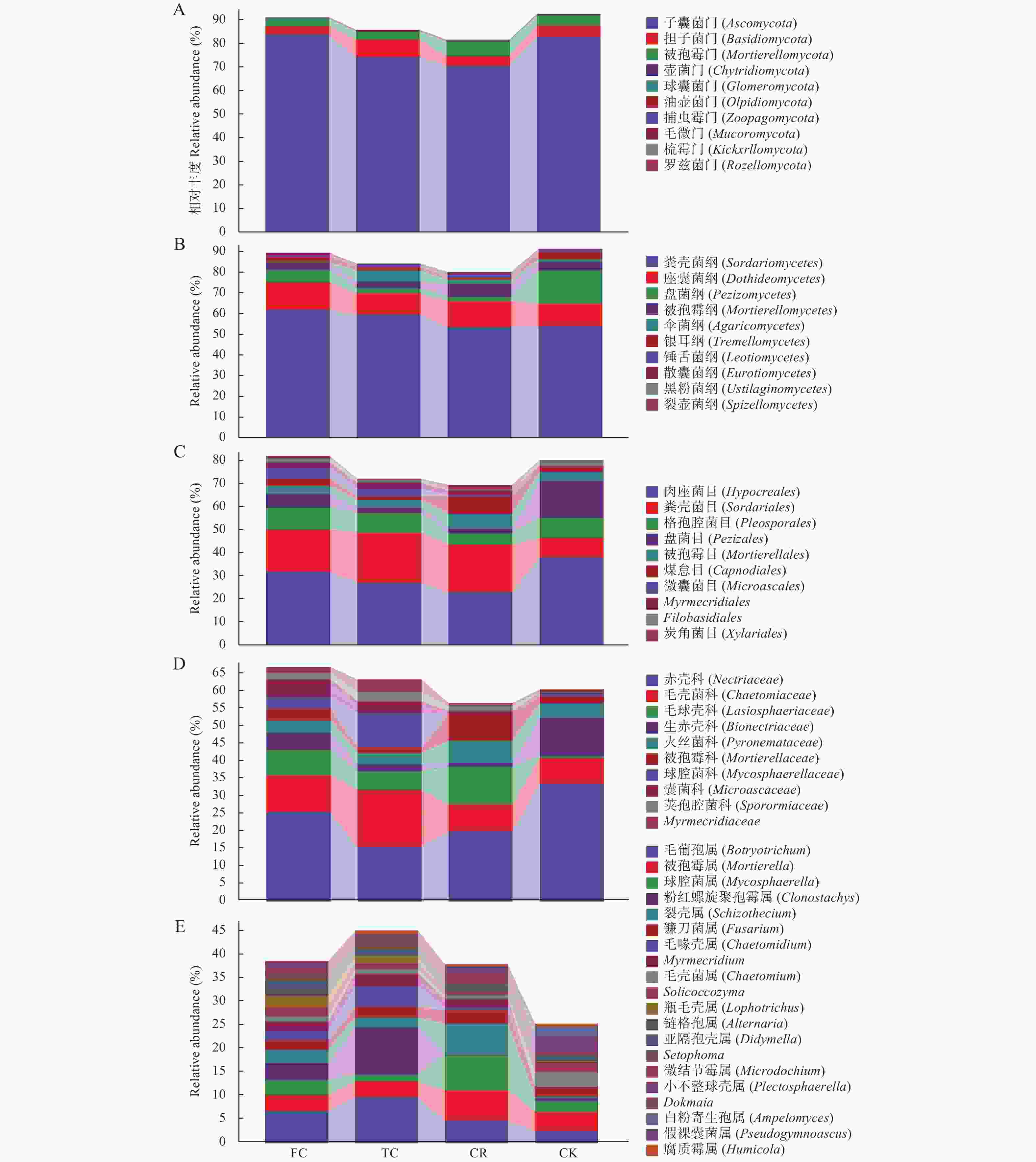
图 3 不同种植模式谷子土壤真菌在不同分类水平的相对丰度比较
A-E表示分类单元在门、纲、目、科、和属水平上的百分比。缩写同图1。A-E indicates the percentage of taxa at the phylum, class, order, family, and genus levels. Abbreviations are the same as those given in Fig. 1.
Figure 3. Relative abundance of soil fungi in foxtail millet under different cropping patterns at different classification levels
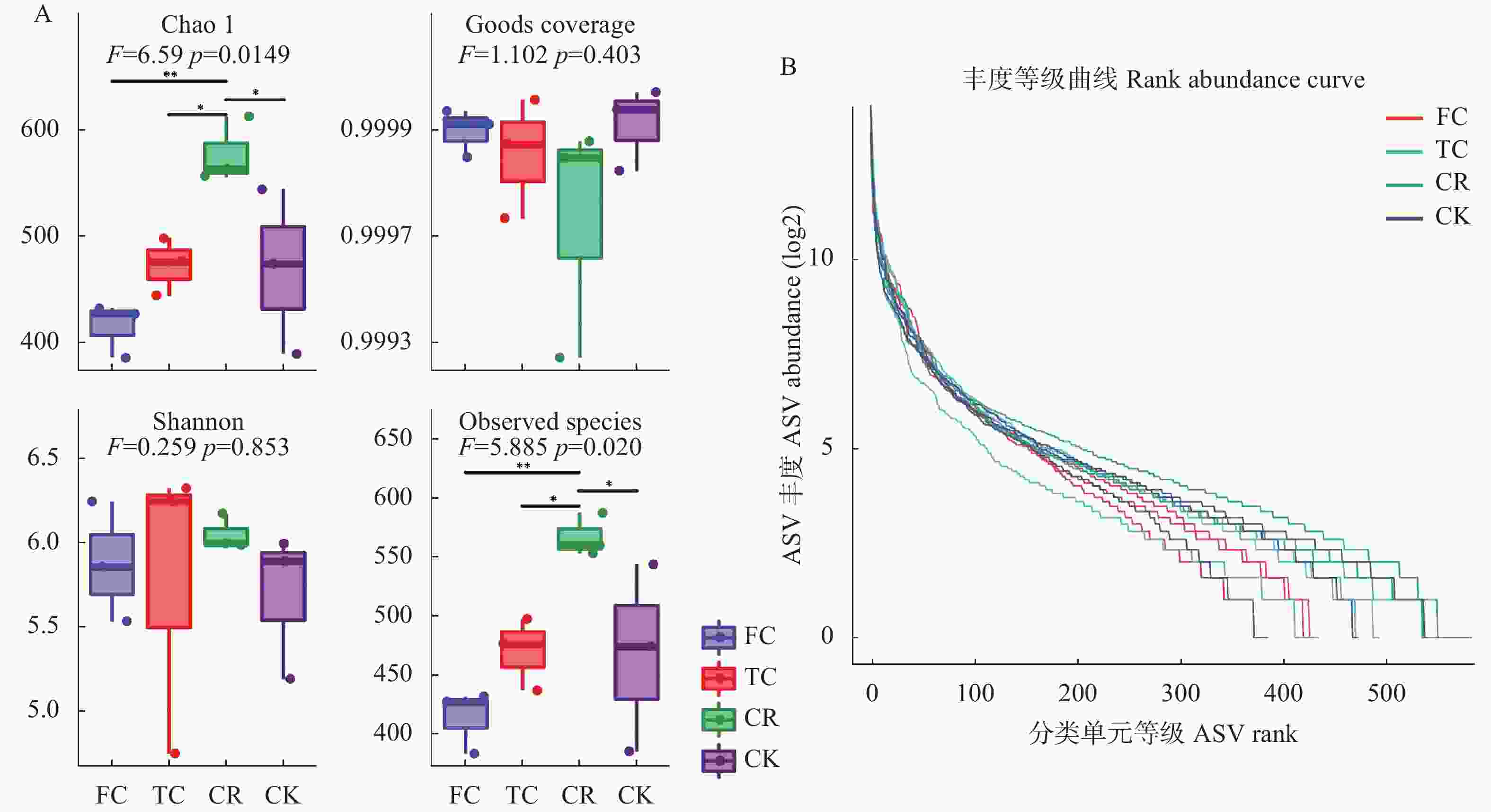
图 4 不同种植模式谷子土壤真菌群落的Alpha多样性分析
A: α多样性指数; B: 丰度等级曲线。CK: 撂荒地; CR: 谷子-玉米轮作; TC: 谷子连作3年; FC: 谷子连作5年。A: α diversity index; B: Abundance grade curve. CK: abandoned land; CR: foxtail millet rotation with maize; TC: continuous cropping for 3 years of foxtail millet; FC: continuous cropping for 5 years of foxtail mille.
Figure 4. Alpha diversity analysis of soil fungi community of foxtail millet under different cropping pattens
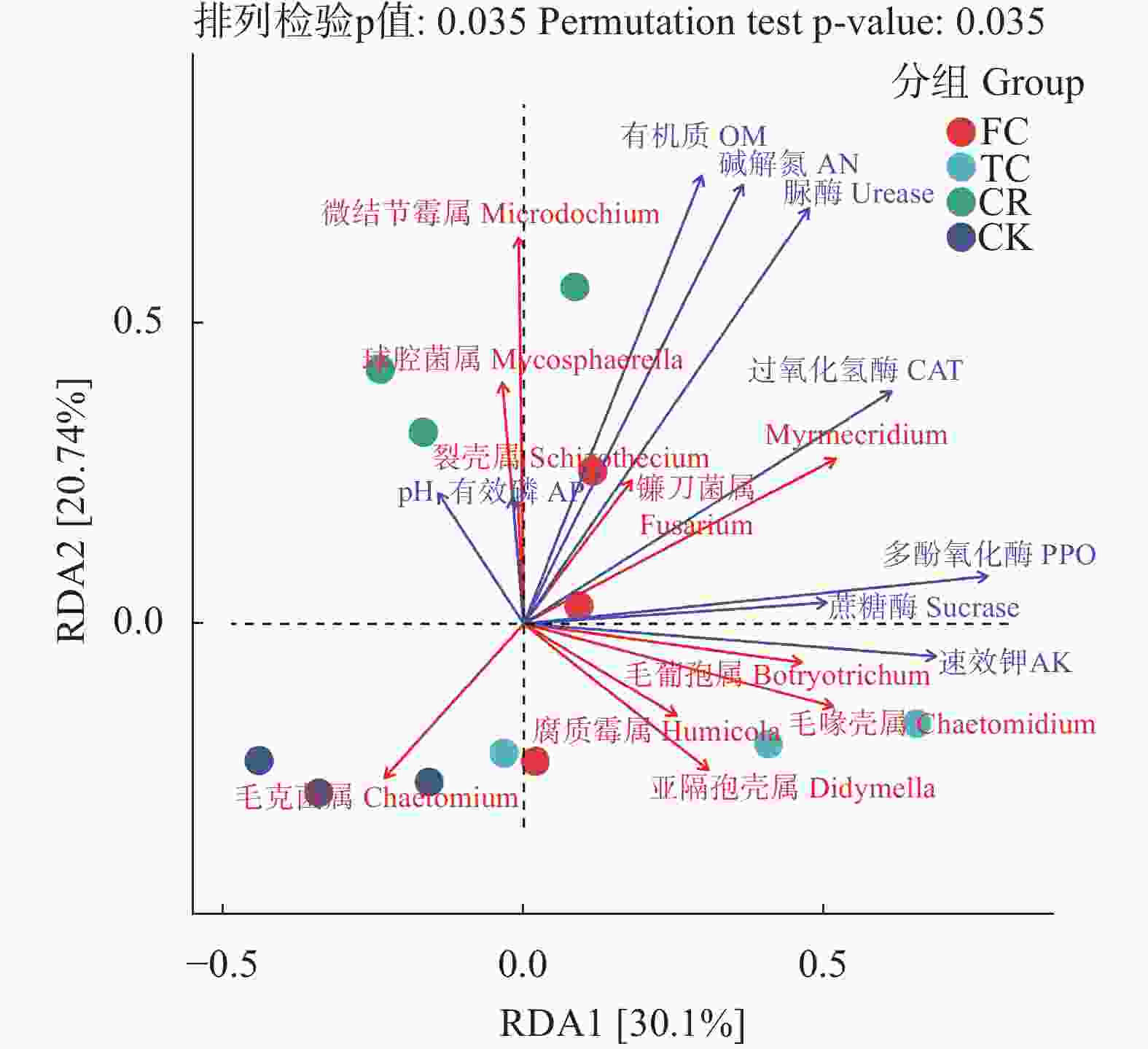
图 6 不同种植模式谷子土壤真菌群落冗余分析
AN: 碱解氮; AK: 速效钾; AP: 有效磷; OM: 有机质; PPO: 多酚氧化酶; CAT: 过氧化氢酶; Urease: 脲酶; Sucrase: 蔗糖酶。CK: 撂荒地; CR: 谷子-玉米轮作; TC: 谷子连作3年; FC: 谷子连作5年。Microdochium:微结节霉属; Mycosphaerella:球腔菌属; Schizothecium:裂壳属; Fusarium:镰刀菌属; Myrmecridium:未找到中文名称; Botryotrichum:毛葡孢属; Humicola:腐质霉属; Chaetomidium:毛喙壳属; Didymella:亚隔孢壳属; Chaetomium:毛壳菌属。AN: Alkaline hydrolysis nitrogen, AK: Rapid available potassium, AP: Rapid available phosphorus, OM: Organic matter, PPO: Polyphenol oxidase, CAT: catalase. CK: abandoned land; CR: foxtail millet rotation with maize; TC: continuous cropping for 3 years of foxtail millet; FC: continuous cropping for 5 years of foxtail mille.
Figure 6. Redundancy analysis of soil fungal community of foxtail millet under different cropping pattens
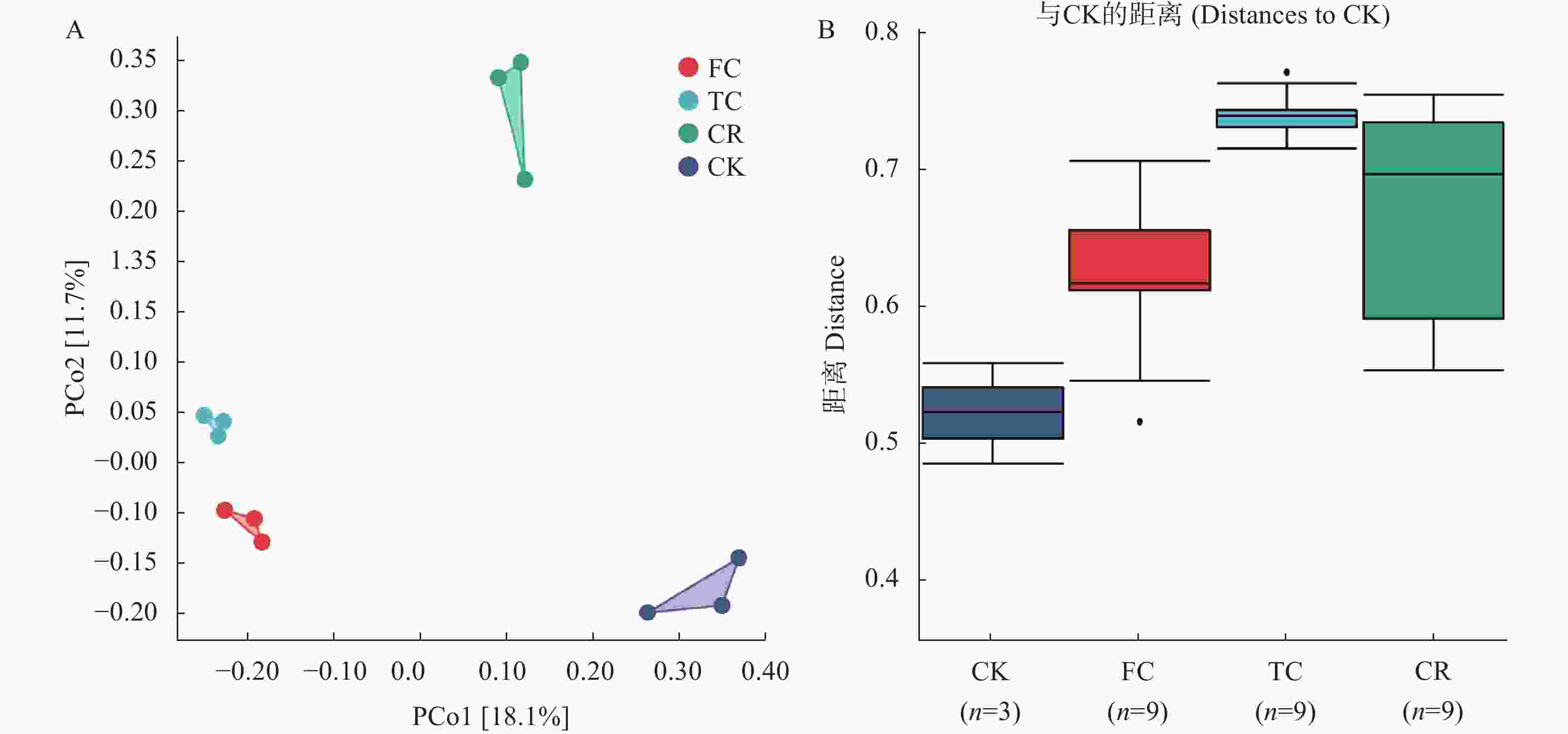
图 5 不同种植模式谷子土壤真菌群落β多样性分析
A: PCoA分析; B: 组间差异分析。CK: 撂荒地; CR: 谷子-玉米轮作; TC: 谷子连作3年; FC: 谷子连作5年。A: PCoA analysis; B: Analysis of differences among four groups. CK: abandoned land; CR: foxtail millet rotation with maize; TC: continuous cropping for 3 years of foxtail millet; FC: continuous cropping for 5 years of foxtail mille.
Figure 5. β diversity analysis of soil fungal community of foxtail millet under different cropping pattens
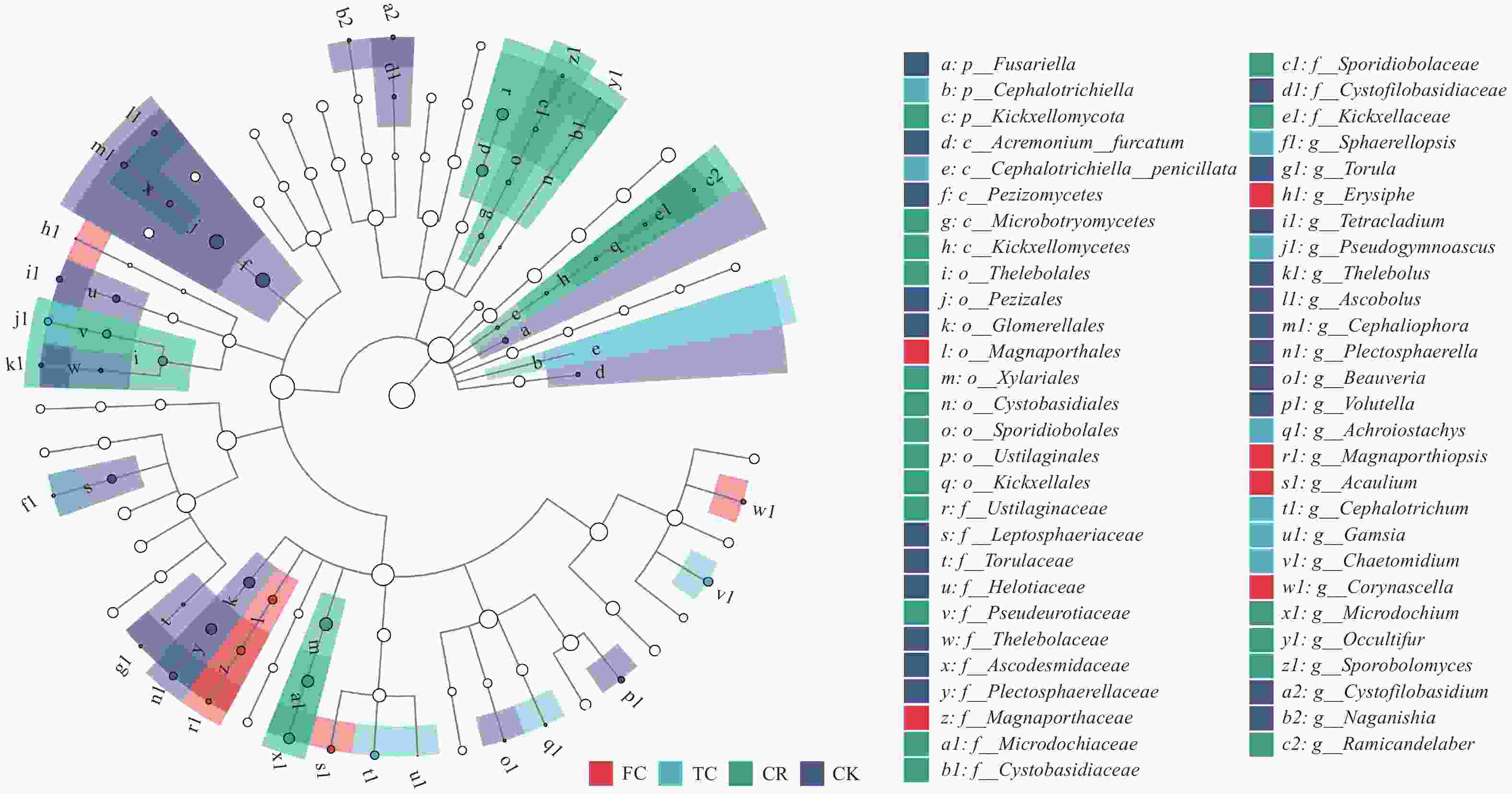
图 7 不同种植模式谷子土壤真菌群落LEfSe分析
由里到外, 依次是门、纲、目、科、属和种水平, 其中实心节点代表在CK、CR、TC和FC中起重要作用的微生物类群, 空心节点表示不起重要作用的物种。CK: 撂荒地; CR: 谷子-玉米轮作; TC: 谷子连作3年; FC: 谷子连作5年。From inside to outside, the species at phylum, class, order, family, genus and species level are successively. Solid nodes represent the microbial groups that play an important role in CK, CR, TC and FC, while hollow nodes represent the species that do not play an important role in the four samples. CK: abandoned land; CR: foxtail millet rotation with maize; TC: continuous cropping for 3 years of foxtail millet; FC: continuous cropping for 5 years of foxtail mille.
Figure 7. LEfSe analysis of soil fungal community of foxtail millet under different cropping pattens
表 1 不同种植模式谷子土壤的真菌高通量测序结果
Table 1. High-throughput sequencing results of fungi in millet soils with different cropping patterns
样本 Sample 去引物 Input 质量过滤 Filtered 去噪 Denoised 拼接 Merged 去嵌合体 Nonchimeric 去 singleton Nonsingleton CK1 84 027 72 625 72 198 71 545 70 622 70 622 CK2 126 381 113 085 112 560 111 845 109 959 109 959 CK3 89 761 78 138 77 541 76 793 75 687 75 687 CR1 191 960 176 283 175 227 174 315 166 205 166 205 CR2 107 110 94 546 93 956 93 200 92 359 92 358 CR3 117 377 102 500 101 873 101 068 98 463 98 463 TC1 137 752 120 634 120 061 119 468 117 184 117 184 TC2 99 344 86 259 85 673 85 041 82 241 82 241 TC3 88 864 78 645 78 247 77 656 77 136 77 136 FC1 116 128 102 142 101 466 100 870 98 526 98 526 FC2 104 460 91 841 91 398 90 841 90 022 90 022 FC3 96 306 84 106 83 632 82 980 82 011 82 011 CK: 撂荒地; CR: 谷子-玉米轮作; TC: 谷子连作3年; FC: 谷子连作5年。各处理缩写后的数据为重复。CK: abandoned land; CR: foxtail millet rotation with maize; TC: continuous cropping for 3 years of foxtail millet; FC: continuous cropping for 5 years of foxtail mille. The data after abbreviation of treatment is the replicate. 表 2 土壤酶活性与理化性质的相关性分析
Table 2. Correlation analysis between soil enzyme activity and physicochemical properties
相关系数
Pearson
CorrelationpH 碱解氮
Alkaline hydrolysis
nitrogen有效磷
Rapid available
phosphorus速效钾
Rapid available
potassium有机质
Organic matter多酚氧化酶
PPO过氧化氢酶
CAT脲酶
urease蔗糖酶
sucrasepH 1 0.466 0.521 0.303 0.314 −0.291 −0.331 −0.203 −0.616* 碱解氮
Alkaline hydrolysis nitrogen0.466 1 0.588* 0.272 0.906** 0.170 0.360 0.628* 0.081 有效磷
Rapid available phosphorus0.521 0.588* 1 0.216 0.614* 0.012 0.024 0.281 0.131 速效钾
Rapid available potassium0.303 0.272 0.216 1 0.147 0.669* 0.451 0.326 0.230 有机质
Organic matter0.314 0.906** 0.614* 0.147 1 0.145 0.367 0.654* 0.175 多酚氧化酶
PPO−0.291 0.170 0.012 0.669* 0.145 1 0.855** 0.681* 0.769** 过氧化氢酶
CAT−0.331 0.360 0.024 0.451 0.367 0.855** 1 0.840** 0.682* 脲酶
urease−0.203 0.628* 0.281 0.326 0.654* 0.681* 0.840** 1 .691* 蔗糖酶 sucrase −0.616* 0.081 0.131 0.230 0.175 0.769** 0.682* 0.691* 1 * 表示在0.05水平差异显著, ** 表示在0.01水平差异显著。* Correlation is significant at the 0.05 level; ** correlation is significant at the 0.01 level. 表 3 土壤真菌群落多样性指数与理化性质、酶活性的相关性分析
Table 3. Correlation analysis of soil fungal community diversity index with physicochemical properties and enzyme activities
相关系数
Pearson CorrelationChao1指数
Chao1 indexGood’s coverage指数
Good’s coverage indexObserved species指数
Observed species indexShannon指数
Shannon indexPH −0.339 0.265 −0.316 −0.108 碱解氮 Alkaline hydrolysis nitrogen −0.038 0.369 0.006 0.024 有效磷 Rapid available phosphorus −0.105 0.381 −0.065 −0.076 速效钾 Rapid available potassium 0.505 0.097 0.539 0.227 有机质 Organic matter −0.048 0.389 −0.002 −0.054 多酚氧化酶 PPO 0.776** −0.398 0.770** 0.038 过氧化氢酶 CAT 0.633* −0.38 0.625* 0.034 脲酶 urease 0.348 −0.021 0.363 −0.088 蔗糖酶 sucrase 0.613* −0.21 0.614* −0.048 * 表示在0.05水平差异显著, ** 表示在0.01水平差异显著。* Correlation is significant at the 0.05 level; ** correlation is significant at the 0.01 level. -
[1] MARTIN L, MESSAGER E, BEDIANASHVILI G, et al. The place of millet in food globalization during Late Prehistory as evidenced by new bioarchaeological data from the Caucasus[J]. Scientific Reports, 2021, 11(1): 13124 doi: 10.1038/s41598-021-92392-9 [2] POSPIESZNY Ł, MAKAROWICZ P, LEWIS J, et al. Isotopic evidence of millet consumption in the Middle Bronze Age of East-Central Europe[J]. Journal of Archaeological Science, 2021, 126: 105292 doi: 10.1016/j.jas.2020.105292 [3] DIAO X M. Production and genetic improvement of minor cereals in China[J]. The Crop Journal, 2017, 5(2): 103−114 doi: 10.1016/j.cj.2016.06.004 [4] HOU D Z, CHEN J, REN X, et al. A whole foxtail millet diet reduces blood pressure in subjects with mild hypertension[J]. Journal of Cereal Science, 2018, 84: 13−19 doi: 10.1016/j.jcs.2018.09.003 [5] 李锐, 刘瑜, 褚贵新, 等. 棉花连作对北疆土壤酶活性、致病菌及拮抗菌多样性的影响[J]. 中国生态农业学报, 2015, 23(4): 432−440LI R, LIU Y, CHU G X, et al. Response of soil enzyme activity and microbial community structure, diversity to continuous cotton cropping in northern Xinjiang[J]. Chinese Journal of Eco-Agriculture, 2015, 23(4): 432−440 [6] XIONG W, LI Z G, LIU H J, et al. The effect of long-term continuous cropping of black pepper on soil bacterial communities as determined by 454 pyrosequencing[J]. PLoS ONE, 2015, 10(8): e0136946 doi: 10.1371/journal.pone.0136946 [7] URASHIMA Y, SONODA T, FUJITA Y, et al. Application of PCR-denaturing-gradient gel electrophoresis (DGGE) method to examine microbial community structure in Asparagus fields with growth inhibition due to continuous cropping[J]. Microbes and Environments, 2011, 27: 43−48 [8] ZHOU X G, GAO D M, LIU J, et al. Changes in rhizosphere soil microbial communities in a continuously monocropped cucumber (Cucumis sativus L.) system[J]. European Journal of Soil Biology, 2014, 60: 1−80 [9] LIU W X, WANG Q L, WANG B Z, et al. Changes in the abundance and structure of bacterial communities under long-term fertilization treatments in a peanut monocropping system[J]. Plant and Soil, 2015, 395(1/2): 415−427 [10] QIN S, YEBOAH S, XU X X, et al. Analysis on fungal diversity in rhizosphere soil of continuous cropping potato subjected to different furrow-ridge mulching managements[J]. Frontiers in Microbiology, 2017, 0: 845 [11] LEI H Y, LIU A K, HOU Q W, et al. Diversity patterns of soil microbial communities in the Sophora flavescens rhizosphere in response to continuous monocropping[J]. BMC Microbiology, 2020, 20(1): 272 doi: 10.1186/s12866-020-01956-8 [12] 侯慧, 董坤, 杨智仙, 等. 连作障碍发生机理研究进展[J]. 土壤, 2016, 48(6): 1068−1076HOU H, DONG K, YANG Z X, et al. Advance in mechanism of continuous cropping obstacle[J]. Soils, 2016, 48(6): 1068−1076 [13] YING Y X, DING W L, ZHOU Y Q, et al. Influence of Pan- axginseng continuous cropping on metabolic function of soil microbial communities[J]. Chinese Herbal Medi- cines, 2012, 4(4): 329−334 [14] 刘建国, 张伟, 李彦斌, 等. 新疆绿洲棉花长期连作对土壤理化性状与土壤酶活性的影响[J]. 中国农业科学, 2009, 42(2): 725−733 doi: 10.3864/j.issn.0578-1752.2009.02.044 [15] LIU J G, ZHANG W, LI Y B, et al. Effects of long-term continuous cropping system of cotton on soil physical-chemical properties and activities of soil enzyme in oasis in Xinjiang[J]. Scientia Agricultura Sinica, 2009, 42(2): 725−733 [16] LI X G, DING C F, HUA K, et al. Soil sickness of peanuts is attributable to modifications in soil microbes induced by peanut root exudates rather than to direct allelopathy[J]. Soil Biology and Biochemistry, 2014, 78: 149−159 doi: 10.1016/j.soilbio.2014.07.019 [17] 张晓玲, 潘振刚, 周晓锋, 等. 自毒作用与连作障碍[J]. 土壤通报, 2007, 38(4): 781−784 doi: 10.3321/j.issn:0564-3945.2007.04.033ZHANG X L, PAN Z G, ZHOU X F, et al. Autotoxicity and Continuous Cropping Obstacles: A Review[J]. Chinese Journal of Soil Science, 2007, 38(4): 781−784 doi: 10.3321/j.issn:0564-3945.2007.04.033 [18] WU Z J, XIE Z K, YANG L, et al. Identification of autotoxins from root exudates of Lanzhou lily (Lilium davidii var unicolor)[J]. Allelopathy Journal, 2015, 35: 35−48 [19] LI X G, DING C F, ZHANG T L, et al. Fungal pathogen accumulation at the expense of plant-beneficial fungi as a consequence of consecutive peanut mono culturing[J]. Soil Biology and Biochemistry, 2014, 72: 11−18 doi: 10.1016/j.soilbio.2014.01.019 [20] LU F G, WANG L, LIU Y N, et al. Analysis of culturable fungal diversity in rhizosphere soil of healthy and diseased cotton in Southern Xinjiang[J]. African Journal of Microbiology Research, 2012, 6: 7357−7364 doi: 10.5897/AJMR12.1146 [21] NAIR A, NGOUAJIO M. Soil microbial biomass, functional microbial diversity, and nematode community structure as affected by cover crops and compost in an organic vegetable production system[J]. Applied Soil Ecology, 2012, 58: 45−55 doi: 10.1016/j.apsoil.2012.03.008 [22] WANG Y, XU J, SHEN J H, et al. Tillage, residue burning and crop rotation alter soil fungal community and water-stable aggregation in arable fields[J]. Soil and Tillage Research, 2010, 107(2): 71−79 doi: 10.1016/j.still.2010.02.008 [23] DE BOER W, FOLMAN L B, SUMMERBELL R C, et al. Living in a fungal world: impact of fungi on soil bacterial niche development[J]. FEMS Microbiology Reviews, 2005, 29(4): 795−811 doi: 10.1016/j.femsre.2004.11.005 [24] SOBEK E, ZAK J. The Soil FungiLog procedure: method and analytical approaches toward understanding fungal functional diversity[J]. Mycologia, 2003, 95: 590−602 doi: 10.1080/15572536.2004.11833063 [25] SONG X H, PAN Y, LI L Y, et al. Composition and diversity of rhizosphere fungal community in Coptis chinensis Franch. continuous cropping fields[J]. PLoS ONE, 2018, 13(3): e0193811 LAHLALI R, IBRAHIM D, BELABESS Z, et al. High-throughput molecular technologies for unraveling the mystery of soil microbial community: challenges and future prospects[J]. Heliyon, 2021, 7(10): e08142 [26] 李夏, 妙佳源, 高小丽, 等. 连作条件下谷子叶片衰老与活性氧代谢研究[J]. 中国农业大学学报, 2016, 21(4): 1−9 doi: 10.11841/j.issn.1007-4333.2016.04.01LI X, MIAO J Y, GAO X L, et al. Leaf senescence and reactive oxygen metabolism of millet under continuous cropping[J]. Journal of China Agricultural University, 2016, 21(4): 1−9 doi: 10.11841/j.issn.1007-4333.2016.04.01 [27] 卢成达, 郭志利, 李阳, 等. 长期定点连作及单序轮作处理对旱地谷子的光合特性、根系构型和产量的影响[J]. 农学学报, 2019, 9(5): 10−14 doi: 10.11923/j.issn.2095-4050.cjas18090004LU C D, GUO Z L, LI Y, et al. Long-term continuous and rotation cropping: effect on photosynthetic characteristics, root configuration and yield of dry -land millet[J]. Journal of Agriculture, 2019, 9(5): 10−14 doi: 10.11923/j.issn.2095-4050.cjas18090004 [28] 妙佳源, 李夏, 周达, 等. 连作对谷子土壤酶活性及养分的影响[J]. 干旱地区农业研究, 2016, 34(3): 123−126,152 doi: 10.7606/j.issn.1000-7601.2016.03.19MIAO J Y, LI X, ZHOU D, et al. Effects of foxtail millet continuous cropping on soil enzyme activities and nutrients[J]. Agricultural Research in the Arid Areas, 2016, 34(3): 123−126,152 doi: 10.7606/j.issn.1000-7601.2016.03.19 [29] DANG K, GONG X W, ZHAO G, et al. Intercropping alters the soil microbial diversity and community to facilitate nitrogen assimilation: a potential mechanism for increasing proso millet grain yield[J]. Frontiers in Microbiology, 2020, 11: 601054 doi: 10.3389/fmicb.2020.601054 [30] DEBENPORT S J, ASSIGBETSE K, BAYALA R, et al. Association of shifting populations in the root zone microbiome of millet with enhanced crop productivity in the Sahel Region (Africa)[J]. Applied and Environmental Microbiology, 2015, 81(8): 2841−2851 doi: 10.1128/AEM.04122-14 [31] 牛倩云, 韩彦莎, 徐丽霞, 等. 作物轮作对谷田土壤理化性质及谷子根际土壤细菌群落的影响[J]. 农业环境科学学报, 2018, 37(12): 2802−2809 doi: 10.11654/jaes.2018-0128NIU Q Y, HAN Y S, XU L X, et al. Effects of crop rotation on soil physicochemical properties and bacterial community of foxtail millet rhizosphere soil[J]. Journal of Agro-Environment Science, 2018, 37(12): 2802−2809 doi: 10.11654/jaes.2018-0128 [32] 孙倩, 吴宏亮, 陈阜, 等. 不同轮作模式下作物根际土壤养分及真菌群落组成特征[J]. 环境科学, 2020, 41(10): 4682−4689SUN Q, WU H L, CHEN F, et al. Characteristics of soil nutrients and fungal community composition in crop rhizosphere under different rotation patterns[J]. Environmental Science, 2020, 41(10): 4682−4689 [33] STRICKLAND M S, ROUSK J. Considering fungal: bacterial dominance in soils — Methods, controls, and ecosystem implications[J]. Soil Biology and Biochemistry, 2010, 42(9): 1385−1395 doi: 10.1016/j.soilbio.2010.05.007 [34] LIU R R, XIAO Z Y, HASHEM A, et al. Mycorrhizal fungal diversity and its relationship with soil properties in Camellia oleifera[J]. Agriculture, 2021, 11: 470 doi: 10.3390/agriculture11060470 [35] CALLAHAN B, MCMURDIE P, ROSEN M J, et al. DADA2: High resolution sample inference from Illumina amplicon data[J]. Nature Methods, 2016, 13: 581−583 doi: 10.1038/nmeth.3869 [36] CHAO A. Nonparametric estimation of the number of classes in a population[J]. Scandinavian Journal of Statistics, 1984, 11: 265−270 [37] RAMETTE A. Multivariate analyses in microbial ecology[J]. FEMS Microbiology Ecology, 2007, 62(2): 142−160 doi: 10.1111/j.1574-6941.2007.00375.x [38] 郝晓芬, 王根全, 郭二虎, 等. 连作、轮作对谷子根际细菌群落结构的影响[J]. 农业环境科学学报, 2022, 41(3): 585−596 doi: 10.11654/jaes.2021-0649HAO X F, WANG G Q, GUO E H, et al. Effects of continuous cropping and rotation on rhizosphere bacterial community structure of millet[J]. Journal of Agro-Environment Science, 2022, 41(3): 585−596 doi: 10.11654/jaes.2021-0649 [39] KANG S M, KHAN A L, WAQAS M, et al. Plant growth-promoting rhizobacteria reduce adverse effects of salinity and osmotic stress by regulating phytohormones and antioxidants in Cucumis sativus[J]. Journal of Plant Interactions, 2014, 9(1): 673−682 doi: 10.1080/17429145.2014.894587 [40] BANNING N, GLEESON D, GRIGG A, et al. Soil microbial community successional patterns during forest ecosystem restoration[J]. Applied and Environmental Microbiology, 2011, 77: 6158−6164 doi: 10.1128/AEM.00764-11 [41] HARRIS J. Soil microbial communities and restoration ecology: facilitators or followers?[J]. Science, 2009, 325(5940): 573−574 doi: 10.1126/science.1172975 [42] KIM Y C, LEVEAU J, GARDENER B M, et al. The multifactorial basis for plant health promotion by plant-associated bacteria[J]. Applied and Environmental Microbiology, 2011, 77(5): 1548−1555 doi: 10.1128/AEM.01867-10 [43] LI X G, DING C F, HUA K, et al. Soil sickness of peanuts is attributable to modifications in soil microbes induced by peanut root exudates rather than to direct allelopathy[J]. Soil Biology and Biochemistry, 2014, 78: 149−159 doi: 10.1016/j.soilbio.2014.07.019 [44] 保丽美, 丁亚芳, 魏云林, 等. 三七连作与休闲土壤真菌群落组成与多样性分析[J]. 中药材, 2021, 44(1): 7−12 [45] ROUSK J, BROOKES P C, BÅÅTH E. Contrasting soil pH effects on fungal and bacterial growth suggest functional redundancy in carbon mineralization[J]. Applied and Environmental Microbiology, 2009, 75(6): 1589−1596 doi: 10.1128/AEM.02775-08 [46] WU H M, QIN X J, WANG J Y, et al. Rhizosphere responses to environmental conditions in Radix pseudostellariae under continuous monoculture regimes[J]. Agriculture, Ecosystems & Environment, 2019, 270/271: 19−31 [47] 孙倩, 吴宏亮, 陈阜, 等. 宁夏中部干旱带不同作物根际土壤真菌群落多样性及群落结构[J]. 微生物学通报, 2019, 46(11): 2963−2972SUN Q, WU H L, CHEN F, et al. Fungal community diversity and structure in rhizosphere soil of different crops in the arid zone of central Ningxia[J]. Microbiology China, 2019, 46(11): 2963−2972 [48] 王海英, 郭守玉, 黄满荣, 等. 子囊菌较担子菌具有更快的进化速率和更高的物种多样性[J]. 中国科学: 生命科学. 2010, 40(8): 731–737WANG H Y, GUO S Y, HUANG M R, et al. Ascomycota has faster evolutionary rate and higher species diversity than Basidiomycota (Fungi). Sci China Life Sci, 2010, 40(8): 731–737 [49] SHE S, NIU J, ZHANG C, et al. Significant relationship between soil bacterial community structure and incidence of bacterial wilt disease under continuous cropping system[J]. Archives of Microbiology, 2017, 199(2): 267−275 doi: 10.1007/s00203-016-1301-x [50] WANG Y Z, XU X M, LIU T M, et al. Analysis of bacterial and fungal communities in continuous-cropping ramie (Boehmeria nivea L. Gaud) fields in different areas in China[J]. Scientific Reports, 2020, 10(1): 3264 doi: 10.1038/s41598-020-58608-0 [51] SHANTHIYAA V, SARAVANAKUMAR D, RAJENDRAN L, et al. Use of chaetomium globosum for biocontrol of potato late blight disease[J]. Crop Prot, 2013, 25: 33−38 [52] 宁琪, 陈林, 李芳, 等. 被孢霉对土壤养分有效性和秸秆降解的影响[J]. 土壤学报, 2022, 59(1): 206−217NING Q, CHEN L, LI F, et al. Effects of Mortierella on nutrient availability and straw decomposition in soil[J]. Acta Pedologica Sinica, 2022, 59(1): 206−217 [53] ZHANG H S, WU X, LI G, et al. Interactions between arbuscular mycorrhizal fungi and phosphate-solubilizing fungus (Mortierella sp. ) and their effects on Kostelelzkya virginica growth and enzyme activities of rhizosphere and bulk soils at different salinities[J]. Biology and Fertility of Soils, 2011, 47: 543−554 doi: 10.1007/s00374-011-0563-3 [54] KOECHLI C, CAMPBELL A N, PEPE-RANNEY C, et al. Assessing fungal contributions to cellulose degradation in soil by using high-throughput stable isotope probing[J]. Soil Biology and Biochemistry, 2019, 130: 150−158 doi: 10.1016/j.soilbio.2018.12.013 [55] LIAO X G, FANG W G, LIN L C, et al. Metarhizium robertsii produces an extracellular invertase (MrINV) that plays a pivotal role in rhizospheric interactions and root colonization[J]. PLoS One, 2013, 8(10): e78118 doi: 10.1371/journal.pone.0078118 [56] 代鹏, 陈海琴, 顾震南, 等. 高山被孢霉生产多不饱和脂肪酸发酵条件的研究进展[J]. 食品工业科技, 2014, 35(5): 354−359DAI P, CHEN H Q, GU Z N, et al. Research progress in fermentation condition for polyunsaturated fatty acids by Mortierella Alpina[J]. Science and Technology of Food Industry, 2014, 35(5): 354−359 [57] 沈钰森, 王建升, 盛小光, 等. 十字花科植物黑斑病的研究进展[J]. 核农学报, 2021, 35(3): 623−634 doi: 10.11869/j.issn.100-8551.2021.03.0623SHEN Y S, WANG J S, SHENG X G, et al. Research progress on black spot in cruciferous plants[J]. Journal of Nuclear Agricultural Sciences, 2021, 35(3): 623−634 doi: 10.11869/j.issn.100-8551.2021.03.0623 [58] 康子腾, 姜黎明, 罗义勇, 等. 植物病原链格孢属真菌的致病机制研究进展[J]. 生命科学, 2013, 25(9): 908−914KANG Z T, JIANG L M, LUO Y Y, et al. The research advances of mechanism of pathogenicity of Alternaria phytopathogenic fungi[J]. Chinese Bulletin of Life Sciences, 2013, 25(9): 908−914 [59] 吴海霞, 袁梦蕾, 江娜, 等. 粉红螺旋聚孢霉高效生防菌株的筛选与评价[J]. 植物保护, 2021, 47(3): 54−60,95WU H X, YUAN M L, JIANG N, et al. Screening and evaluation of highly efficient biocontrol strains of Clonostachys rosea[J]. Plant Protection, 2021, 47(3): 54−60,95 [60] 刘硕, 郑金柱, 张兆霞, 等. 粉红螺旋聚孢霉对两种林果枝干病原菌的生防作用研究[J]. 山东农业大学学报(自然科学版), 2019, 50(1): 49−51LIU S, ZHENG J Z, ZHANG Z X, et al. Biocontrol activity of Clonostachys rosea against fungal pathogens of forest and fruit trees Botryosphaeria dothidea and Valsa mali[J]. Journal of Shandong Agricultural University (Natural Science Edition), 2019, 50(1): 49−51 [61] GONG C, LIU Y, LIU S Y, et al. Analysis of Clonostachys rosea-induced resistance to grey mould disease and identification of the key proteins induced in tomato fruit[J]. Postharvest Biology and Technology, 2017, 123: 83−93 doi: 10.1016/j.postharvbio.2016.08.004 [62] BORGES Á V, SARAIVA R M, MAFFIA L A. Biocontrol of gray mold in tomato plants by Clonostachys rosea[J]. Tropical Plant Pathology, 2015, 40(2): 71−76 doi: 10.1007/s40858-015-0010-3 [63] LIU Z X, LIU J J, YU Z H, et al. Long-term continuous cropping of soybean is comparable to crop rotation in mediating microbial abundance, diversity and community composition[J]. Soil and Tillage Research, 2020, 197: 104503 doi: 10.1016/j.still.2019.104503 [64] 李春格, 李晓鸣, 王敬国. 大豆连作对土体和根际微生物群落功能的影响[J]. 生态学报, 2006, 26(4): 1144−1150LI C G, LI X M, WANG J G. Effect of soybean continuous cropping on bulk and rhizosphere soil microbial community function[J]. Acta Ecologica Sinica, 2006, 26(4): 1144−1150 [65] 王娟英, 许佳慧, 吴林坤, 等. 不同连作年限怀牛膝根际土壤理化性质及微生物多样性[J]. 生态学报, 2017, 37(17): 5621−5629WANG J Y, XU J H, WU L K, et al. Analysis of physicochemical properties and microbial diversity in rhizosphere soil of Achyranthes bidentata under different cropping years[J]. Acta Ecologica Sinica, 2017, 37(17): 5621−5629 -





 下载:
下载: